SuperClear® Centrifuge Tubes
We designed our SuperClear® high performance centrifuge tubes to successfully meet your needs every time you use them. We test every lot of these tubes at a much higher g-force than we rate them to ensure that you can spin them safely. Our quality control team tests the seals with chloroform. So these don't just look like centrifuge tubes, they really are.
More Value. Not just another disposable centrifuge tube.

SuperClear® tubes are not only better when you use them, the process used to make them is better, helping reduce their environmental and social footprint. And unlike other brands of centrifuge tubes SuperClear® tubes with elastomeric sealing flat caps can be cleaned and re-used.
SuperClear® Tubes have exclusive features and are patented worldwide. The three color tube labeling graphics are copyright and trademark of Labcon.
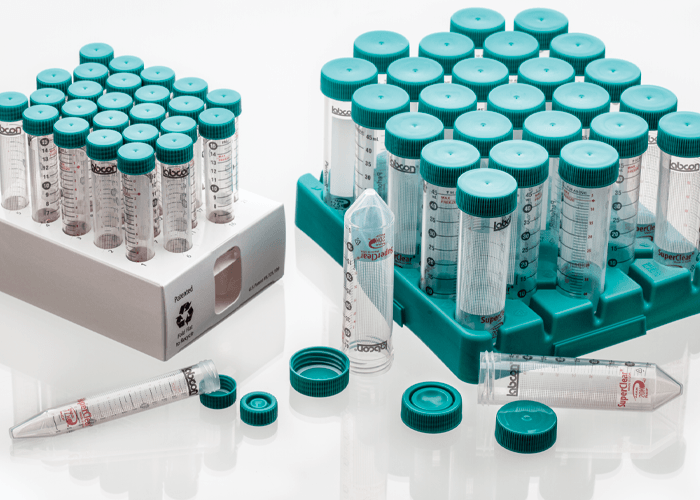

Real tubes, real safety, real performance, real sustainability
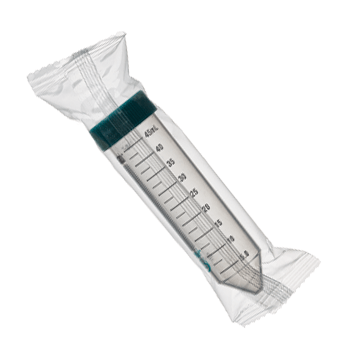
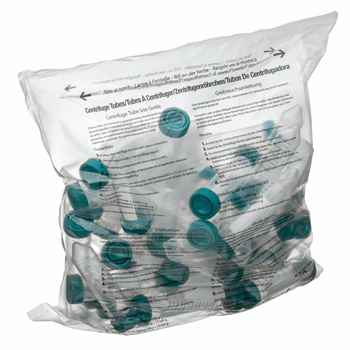
SuperClear® tubes are available in packaging styles for all your needs, including our exclusive IntegraPack® 10 tube sterile packs, individually wrapped sterile packs, reusable polypropylene racks, Patented fiber racks, sterile bulk bags, and non-sterile bulk bags. All our sterile products use medical grade packaging and are radiation sterilized following a validated ISO 11137 method to a sterility assurance level of 10-6.
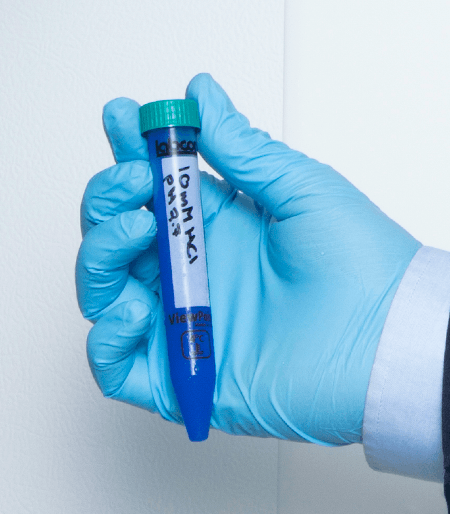
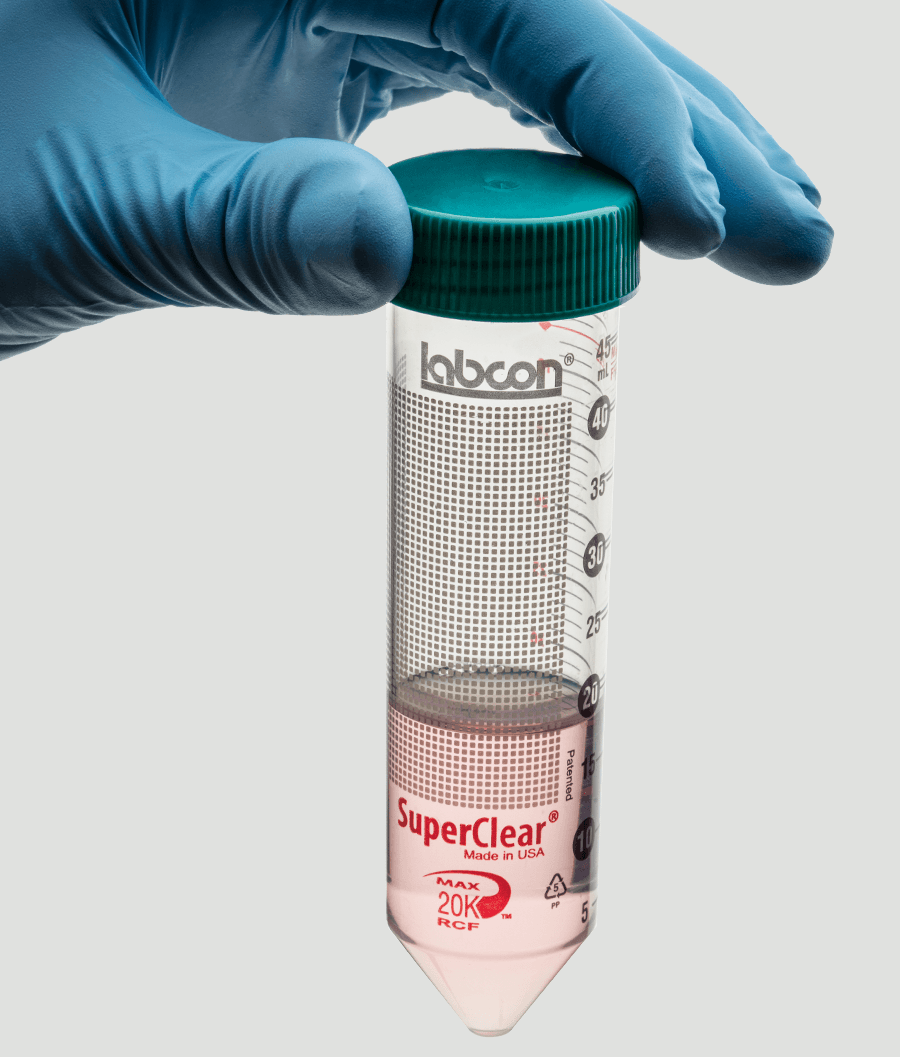
SuperClear® tubes offer you a standard of performance not available from other brands of centrifuge tubes. These tubes are made from a unique medical grade resin that is far more durable than commodity grade resins used in most popular brands of tubes. This high quality medical grade resin makes it possible for you to spin these tubes at g-forces up to 233% greater than other tubes. So you can finally do those extractions you did in polycarbonate tubes before. SuperClear® tubes feature our Patented labeling spot that helps reduce smearing of lab markers at the same time it protects sample labeling from damage during storage and centrifuging.
Unlike other tubes our SuperClear® tubes with our exclusive elastomeric seal are tested and approved to the IATA 95kPa standard for transport of infectious agents. You can be sure your samples will arrive securely when you use SuperClear® tubes.
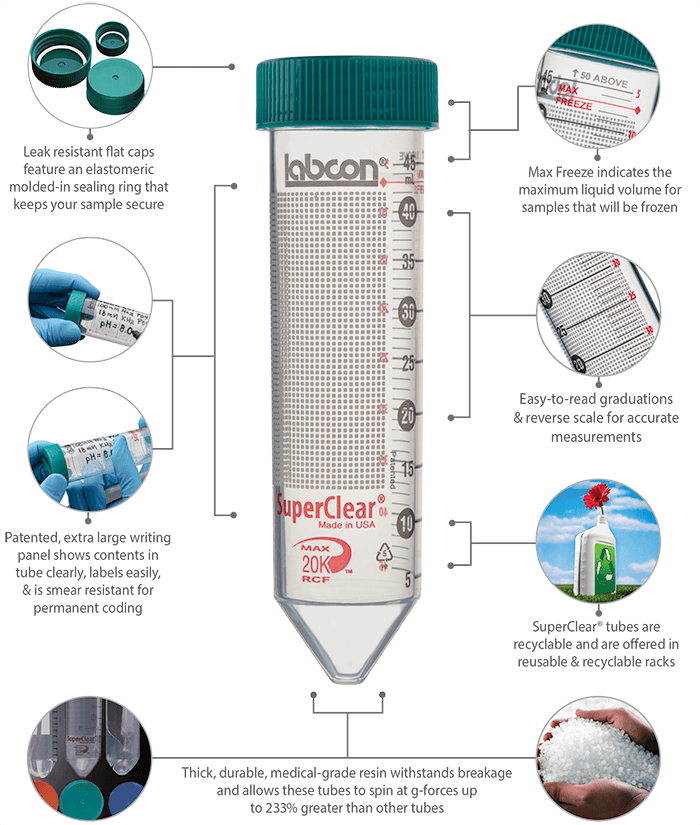
You can order these tubes with a flat cap that includes our exclusive SuperSeal™ integral molded-in elastomeric sealing ring, or a plug style cap that includes a very deep sealing area. The flat style caps can be easily labeled and are designed to allow you to seal them to an evaporation seal with ¾ turn for one-handed operation. And then a final ¼ turn will seal them liquid tight.
SuperClear® tubes are available in packaging styles for all your needs, including our exclusive IntegraPack® 10 tube sterile packs, individually wrapped sterile packs, reusable polypropylene racks, Patented fiber racks, sterile bulk bags, and non-sterile bulk bags. All our sterile products use medical grade packaging and are radiation sterilized following a validated ISO 11137 method.
.| Endotoxin Free (Non pyrogenic) Product samples are exposed to endotoxin-free water and the resulting extraction fluid is tested for contamination using the kinetic turbidimetric Limulus Amoebocyte Lysate (LAL) assay protocol and USP guidelines. All products tested must display less than 0.05 EU/ml to be certified free of endotoxin. |
|
| Nuclease Free (RNase/DNase) Product samples are exposed to nuclease-free water and the resulting extraction fluid is tested for nuclease activity on commercially available 7.5 kb Poly(A) tailed RNA (1µg) and HindIII-digested DNA (1µg) with a one hour 37°C incubation in appropriate buffers. Results are visualized on an agarose gel with appropriate positive and negative controls. Extraction fluid samples must show no degradation of the nucleic acids by the extraction fluid has occurred for the product to be certified as RNase-free and DNase-free |
|
| Adenosine Triphosphate (ATP) Product sample surfaces are tested for the presence of adenosine triphosphate (ATP) using a controlled bioluminescence reaction to detect contamination. Luminescence data is compared to results generated by ATP-free surfaces and surfaces with known amounts of ATP as a positive control. The relative light units result must indicate less than 2 X 10-12 mg/µl of ATP for the product to be certified as ATP free. |
|
| Heavy Metals Free Heavy metals have been tested for using the prescribed USP method and confirmed to have levels lower than 1 part per million (1ppm) |
|
| Validated Sterilization Labcon products are sterilized by radiation sterilization within a dose range of 12-35 kGy. The dose range necessary for the stated sterility assurance level is continuously audited through quarterly bio-burden and sterility validation studies performed according to the ANSI/AAMI/ISO 11137 standard by an independent laboratory. This dosage is sufficient to guarantee a sterility assurance level of 10-6. |
|
| IATA 95kPa Products are pressure and temperature tested to confirm that they are suitable for use as shipping tubes. This work was performed by a third party laboratory. Tubes were filled with a glycerol-water mix and incubated at 25°C in a vacuum chamber at -95kPa for 30 minutes, with no leakage of liquid content observed. Tubes were then incubated at -40°C for two hours at the -95kPA differential, again with no leaking of liquid contents observed. The tubes were then incubated at +55°C for two hours at the -95kPA differential, again with no leaking observed. |
| Bovine Spongiform Encephalopathy-Transmissible Spongiform Encephalopathy These products contain resins that are processed under conditions proven to exceed the European Union standard as listed in the 22nd Commission Directive EC 98/16/EC of March 5th, 1998 as annexed to council Directive EC 76/768/EEC and further Amendment 419 Annex II of 12 June 2001. |
|
| Medical Grade (USP) U.S Pharmacoepia Methods and Guidelines (U.S.P Class VI) are used if applicable. We only use medical grade resins and pre-test all resins for contaminants prior to use. Resins are compliant with FDA CFR title 21-177.1520, 178.3295, 178.3297. |
|
| California Prop 65 No Labcon manufactured disposables contain any of the “listed chemicals” as referenced in the California Safe Drinking Water and Toxic Enforcement Act of 1986, (Prop 65) as revised May 25, 2018. |
|
| Phthalates & Oleamide All our resins are medical grade and are certified free of Bisphenol A (BPA), Oleamide, DiHEMDA, and Phthalates. |
|
| Substances of Very High Concern & REACH All Labcon products are compliant with RoHS Directive 2002/95/EC/-2011/65-2015/863, are free of Substances of Very High Concern (SVHC), and are EU REACH Regulation (EC) No 1907/2006 compliant. |
|
| Origin These products are Made in USA and all components meet the requirements for US origin under the NAFTA agreement. |
| U.S. FDA Registered Labcon is a U.S. FDA registered medical device manufacturer. Our facility is registered by the U.S. government to comply with CFR21 GMP regulations for manufacturing medical devices. |
|
| CE Compliant As applicable Labcon products meet the requirements for CE marking under regulation 2017/746. |
|
| ISO 9001 Quality Registration Labcon has been registered to the ISO 9001 quality management system standar since 1997 and maintains registration to ISO 9001:2015. |
| Material | 100% Virgin Polypropylene |
| Grade | Medical |
| Rating | USP Class VI |
| Colors | Natural (Clear) with Teal or Purple Cap |
| Length | Various |
| Fits | Most popular centrifuges |
| Approved Use | Medical, Research, Industrial, Food & Beverage |
| Lot Expiration | 6 years |
| Sterile Expiration | 5 years |
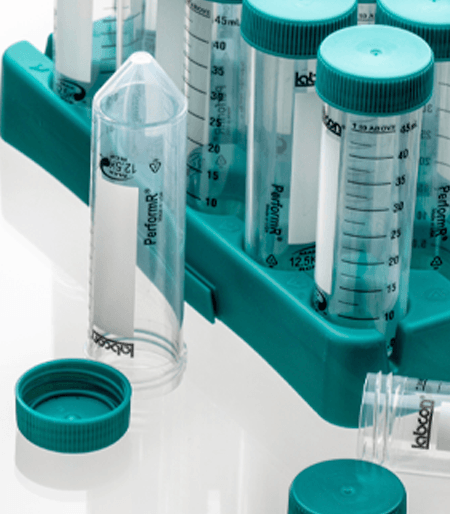
Fiber Rack
These patented collapsable recyclable fiber racks hold 25 tubes and can be recycled like standard cardboard. The clean SBS material is printed with numbers at each well for ease of use. A view port in the ends allows you to see the fluid volume when filling one tube. Available sterilized by irradiation.
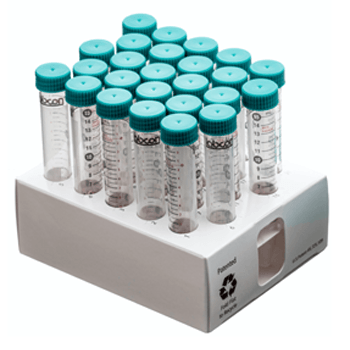
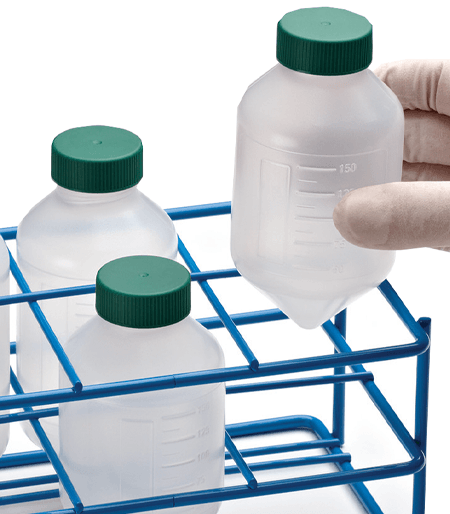
Reusable Rack
This reusable polypropylene rack holds 25 tubes and can be frozen and autoclaved. Perfect for holding any Labcon brand centrifuge tubes. This is a sustainable choice and helps reduce your dependence on wasteful polystyrene foam. Each rack can be connnected to another rack to form a work station.