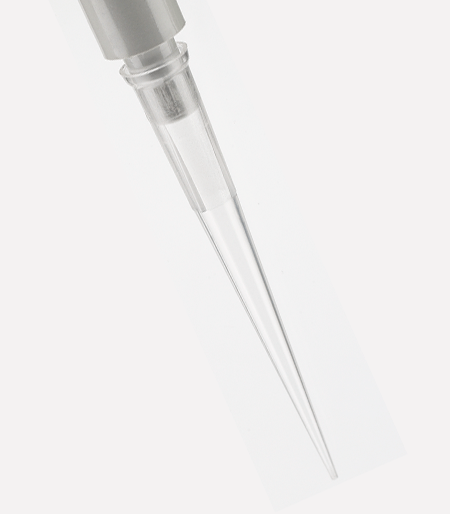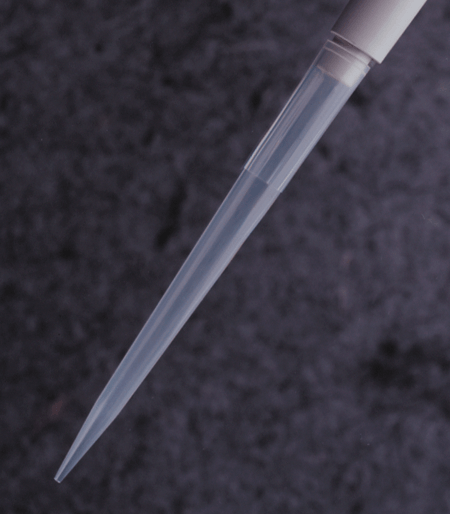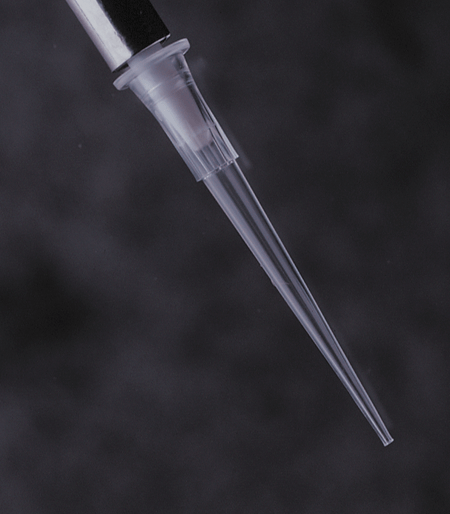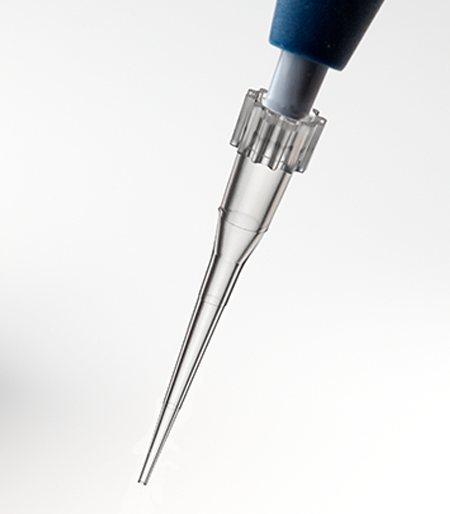
APT™ Automation Pipette Tips
Labcon APT™ pipette tips for automated workstations are designed to work as a plug and play solution on factory specification units.
Available in styles for Beckman® FX and NX and Perkin Elmer® Janus® workstations. 20µL, 30µL, 50µL, and 250µL sizes are available depending upon workstation. All APT™ tips are in a heavy reusable polypropylene SBS footprint rack.


Validated Sterilization
Our APT™ tips in sterile racks, like all our other products, have received valid medical grade sterilization processing compliant with ISO 11137 to an SAL of 10-6. This is a feature you won't get from most pipette tip suppliers, a truly sterile product. Most pipette tip makers offer only a non-validated "presterilized" product.
APT™ Automation Pipette Tips have been designed to fit Beckman® FX, NX and Perkin Elmer® automated pipetting workstations. They are available in 20µL, 30µL, 50µL, and 250µL fluid sizes depending upon the pipetting head.
Each size of these tips is provided in a heavy duty rack designed to accept the heavy load of automated pipette workstations. A color coded platform ensures the correct tip volume can be identfied in the Beckman® unit.
APT™ tips are available in both sterile and non sterile versions. As with all Labcon products the sterile versions are sterilized using a medical grade validation to an SAL of 10-6 to ensure they can be used for diagnostic as well as research applications.
| Endotoxin Free (Non pyrogenic) Product samples are exposed to endotoxin-free water and the resulting extraction fluid is tested for contamination using the kinetic turbidimetric Limulus Amoebocyte Lysate (LAL) assay protocol and USP guidelines. All products tested must display less than 0.05 EU/ml to be certified free of endotoxin. |
|
| Nuclease Free (RNase/DNase) Product samples are exposed to nuclease-free water and the resulting extraction fluid is tested for nuclease activity on commercially available 7.5 kb Poly(A) tailed RNA (1µg) and HindIII-digested DNA (1µg) with a one hour 37°C incubation in appropriate buffers. Results are visualized on an agarose gel with appropriate positive and negative controls. Extraction fluid samples must show no degradation of the nucleic acids by the extraction fluid has occurred for the product to be certified as RNase-free and DNase-free |
|
| Adenosine Triphosphate (ATP) Product sample surfaces are tested for the presence of adenosine triphosphate (ATP) using a controlled bioluminescence reaction to detect contamination. Luminescence data is compared to results generated by ATP-free surfaces and surfaces with known amounts of ATP as a positive control. The relative light units result must indicate less than 2 X 10-12 mg/µl of ATP for the product to be certified as ATP free. |
|
| DNA Free (Human) To confirm the freedom from DNA amplification products are examined by Agarose gel electrophoresis with appropriate positive and negative controls. PCR reactions using extraction fluid must show no PCR- amplified product compared to a positive control containing one picogram of human genomic DNA for the product to be certified as DNA-free. |
|
| Protease Free Product samples are tested and certified to be free of detectable Protease. Extraction fluid is tested for the presence of protease activity by examining test protein degradation in the extraction fluid compared to a negative control and positive control reactions supplemented with 2 nanograms of Proteinase K or Trypsin. |
|
| Heavy Metals Free Heavy metals have been tested for using the prescribed USP method and confirmed to have levels lower than 1 part per million (1ppm) |
|
| Validated Sterilization Labcon products are sterilized by radiation sterilization within a dose range of 12-35 kGy. The dose range necessary for the stated sterility assurance level is continuously audited through quarterly bio-burden and sterility validation studies performed according to the ANSI/AAMI/ISO 11137 standard by an independent laboratory. This dosage is sufficient to guarantee a sterility assurance level of 10-6. |
| Bovine Spongiform Encephalopathy-Transmissible Spongiform Encephalopathy These products contain resins that are processed under conditions proven to exceed the European Union standard as listed in the 22nd Commission Directive EC 98/16/EC of March 5th, 1998 as annexed to council Directive EC 76/768/EEC and further Amendment 419 Annex II of 12 June 2001. |
|
| Medical Grade (USP) U.S Pharmacoepia Methods and Guidelines (U.S.P Class VI) are used if applicable. We only use medical grade resins and pre-test all resins for contaminants prior to use. Resins are compliant with FDA CFR title 21-177.1520, 178.3295, 178.3297. |
|
| California Prop 65 No Labcon manufactured disposables contain any of the “listed chemicals” as referenced in the California Safe Drinking Water and Toxic Enforcement Act of 1986, (Prop 65) as revised May 25, 2018. |
|
| Phthalates & Oleamide All our resins are medical grade and are certified free of Bisphenol A (BPA), Oleamide, DiHEMDA, and Phthalates. |
|
| Substances of Very High Concern & REACH All Labcon products are compliant with RoHS Directive 2002/95/EC/-2011/65-2015/863, are free of Substances of Very High Concern (SVHC), and are EU REACH Regulation (EC) No 1907/2006 compliant. |
|
| Origin These products are Made in USA and all components meet the requirements for US origin under the NAFTA agreement. |
| U.S. FDA Registered Labcon is a U.S. FDA registered medical device manufacturer. Our facility is registered by the U.S. government to comply with CFR21 GMP regulations for manufacturing medical devices. |
|
| CE Compliant As applicable Labcon products meet the requirements for CE marking under regulation 2017/746. |
|
| ISO 9001 Quality Registration Labcon has been registered to the ISO 9001 quality management system standar since 1997 and maintains registration to ISO 9001:2015. |
| Material | 100% Virgin Polypropylene |
| Grade | Medical |
| Rating | USP Class VI |
| Color | Magenta or Natural |
| Length | Various |
| Fits | Beckman® FX and NX or Perkin Elmer® Janus® |
| Approved Use | Medical, Research, Industrial, Food & Beverage |
| Lot Expiration | 6 years |
| Sterile Expiration | 5 years |